The ability to scale up reactions quickly and reliably is a key requirement for pharmaceutical and fine or specialty chemical companies. The Alfa Laval flow reactor technology is a scale-up solution of the future for a wide range of chemical processes.
The Author: Dr. Barry Johnston Products & Applications Mgr., Alfa Laval Reactor Technology
Different companies require different stages and different extents of scale-up. Reactions may be transferred from medicinal chemistry to process R&D, where the quantities are increased from milligrams to grams, or from pilot plant to production scale, where tens or even hundreds of kilograms need to be produced per hour. Performing this scale-up in traditional process development increases the size of the equipment, usually significantly – from laboratory to plant the vessel dimensions may change from a few centimetres to one metre. In continuous flow reactor implementations the laboratory and production dimensions are much smaller and closely toleranced, minimising the decrease in performance and making the steps easier.
The examples presented in this article show how Alfa Laval plate reactors have been used in a range of sizes and environments, from industrial manufacturing to GMP. In the majority of cases, it was possible to transfer the process directly without modifying the conditions or the operating strategy, but where cooling limits are approached with larger scales, alternative strategies were required.
Example from the fine chemicals sector
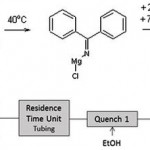
Sigma Aldrich were among the first to demonstrate scale-up from a microreactor to an Alfa Laval ART plate reactor. 2-benzoyl pyridine is an important building block in synthetic chemistry, with an annual demand of about 15 t. It is synthesised by means of a Grignard reaction (Figure 1), which takes less than a minute. The reaction is followed by two online quench steps to obtain a two-phase, solids-free mixture which separates well. The solvent is evaporated from the aqueous phase with a yield of 98 %, thereby eliminating the distillation step that is essential to purify lower-quality batch products.
The flow process was first developed using a glass microreactor (channel area = 0.25 mm2, volume = 2 ml), which had to be operated with a tubing coil to increase the residence time and was capable of producing up to 1 kg of 2-benzoyl pyridine per day.
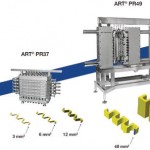
In the pilot study for transferring a micro scale process to production scale, this reaction was applied to operation in a stainless steel Alfa Laval ART PR37 plate reactor (channel area = 3 mm2, reactor volume = 26 ml). Operation in this larger reactor under the same conditions as in the microreactor resulted in an equivalent product. The reaction step, residence time and two quench steps were all combined into the single PR37 unit.
The plate reactor, however, allows operation at even higher throughputs (and at higher flow velocities which result in better mixing and heat transfer performance); if the number of plates were to be increased to ten, 14 kg could be produced per hour. Continuous operation of this unit over a couple of months would equate to the full annual requirement from a reactor that occupies only 30 x 50 cm of bench space. So plate reactors have some simple, pragmatic advantages over traditional batch vessels: they take up less room on the factory floor yet can still be used to make large quantities of product.
Microwave heated syntheses
A further instance of the benefits generated by the superior heating capabilities of small flow channels is when microwave chemistry is scaled up. AstraZeneca has published examples where syntheses developed in microwave ovens have been transferred to flow plate reactors. This is essentially a conversion from batch to continuous simultaneously with the process scale-up.
The advantages of this methodology are that it allows the initial development of a batch synthesis route with very small quantities of material and avoids the problems normally associated with scaling up microwave heating – such as a short penetration depth (the distance a microwave can travel into a standard sample), which limits the size of the reactor. In addition, these rapid heating rates can be achieved with any solvent.
In a specific project reaction there were two competing reaction paths: the cleavage of the carbon-sulphur bond was favoured at low temperatures, while at high temperatures the desired ring closure was promoted.
In batch production it was possible to preheat the individual reactants to +70 °C, then mix them and continue heating the pressure vessel to +110 °C over a period of four minutes; the yield was only 23 %, however. When the reaction was performed inside a microwave oven in a pressure vial (20 ml), the mixture could be heated from +20 to +110 °C in 20 s with an acceptable yield of 56 % – but this was the maximum size for this equipment.
In an ART PR37 plate reactor (channel area = 3 mm2, reactor volume = 100 ml) the reactants were added at the first two ports of the first reaction plate; they reached the reactor temperature (+110 °C) within 10 s at a total flow rate of about 25 ml/min (heating rates of 20 to 30 °C/s are feasible). Within an hour of operation a yield of 59 % was achieved, with productivity at least an order of magnitude greater than with the microwave oven apparatus.
Modular platform
Alfa Laval ART plate reactors are a flexible reactor platform developed to support process intensification and technology conversion (from batch to continuous operation). The modularity of the design means that plates with different channel sizes can be assembled in the same frame to best fit the reaction: process plates are generally connected in series in order to achieve the required residence time.
The benefits of tailoring the reactor configuration to fit the reaction (rather than the reverse as is the case with traditional batch reactors) include improved yield and enhanced safety. The plate reactor range also allows users to scale up the process by selecting a larger unit with similar characteristics that reduces the risks and time required.
Reaching production scale
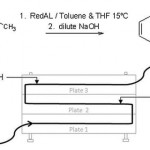
Scale up across the range of plate reactors is illustrated with a prototypical reaction – the reduction of a carbonyl group using an alu-minium hydride reagent. Processes involving organometallic reagents are frequently presented as laboratory demonstrations of flow chemistry. When this reaction was carried out in a PR37 plate reactor (channel area = 3 mm2, reactor volume = 40 ml, Figure 3), the result was a quantitative conversion of aldehyde to alcohol with 0.7 kg/h of product. Both steps – the adduct formation and its subsequent hydrolysis to release the alcohol product – were incorporated in a single reactor that was operated at room temperature.
This process was scaled up in two stages by a factor of 22 using increasingly large plate reactors while maintaining 100 % yield, although far greater increases are also possible utilising Alfa Laval plate reactor technology.
Scale-up stage 1 (channel area increased to 12 mm2, reactor volume = 192 ml): This larger PR37 was operated in the Alfa Laval Atex facility. Reagents were supplied by microgear pumps for the main reactants and HPLC pumps for the quench solution, with a heater-chiller unit to set the reactor temperature.
The ports along the reactor plates allow thermocouples to be fitted in order to show the temperature profile along the reactor. An analysis of the hydrolysed stream showed that no by-products were formed and the production of alcohol was 3 kg/h, though optimising the reactor configuration and operating conditions (high flow rates) could have resulted in over 7 kg/h from the same equipment.
Scale-up stage 2 (channel area increased to 48 mm2, reactor volume = 2.25 l): a PR49 reactor was necessary to achieve higher production rates, although this reactor could be operated with all the same auxiliary equipment as in the PR37 tests. An insulator plate was installed between the reactor plates to allow different temperature zones to be established within the same reactor. The main reaction was carried out at a lower temperature to compensate for the smaller cooling surface area. Different feed and reactor configurations were tested, but once again a GC analysis showed that no by-products were formed.
Operation with a single (two) benzyl acetone feed point corresponds to a total mass flow rate through the PR49 reactor of 55 (78) kg/h, resulting in the production of 11 (15) kg/h of alcohol. It was subsequently calculated that a full, ten-plate PR49 reactor could manufacture 40 kg/h of alcohol – a scale-up factor of 60 compared to the initial PR37 tests.
The above production levels were achieved based on a simple scale-up of the reactor channel area, facilitated by the fact that switching from a large and slow batch process to a faster and smaller plate reactor process eliminated by-product formation and improved the temperature control of the reaction.
Both the stage 1 and stage 2 scale-ups required only a few hours of operation to establish and demonstrate a stable operating condition with the final PR49 set-up yielding >15 kg/h of alcohol product. At the same time, the tests showed how a plate reactor production unit can be operated in a safe and reliable manner.
Support for testing flow technology
In addition to supplying plate reactors, Alfa Laval reactor technology offers process evaluation and consultancy services. Customers can test their specific chemistry with Alfa Laval ART plate reactors, supported by process experts, either on their own premises or at Alfa Laval’s site near Stockholm, Sweden. When a customer invests in this equipment, Alfa Laval provides start-up assistance, troubleshooting, process optimisation and technical training by experts from the Alfa Laval reactor technology team. Spare parts are available at short notice.
Hall 4.0, Booth D4
cpp-net.com/0212433
Batch vs. flow
Heat transfer
The success of the Sigma Aldrich case is underpinned by the heat transfer capabilities of small channels. The reaction was carried out at high concentrations, which meant high rates of heat release from this exothermic reaction. The surface area available in a reactor for cooling (or heating) is a critical component for scale-up and safe process development. Whereas a batch vessel may have a few square metres per cubic metre of reactant volume, millimetre scale channels in flow reactors can have a surface area of hundreds of square metres per cubic metre. The greater the cooling capacity of the reactor (which is proportional to the heat transfer area x the heat transfer coefficient), the higher the reaction rates which can be accommodated and the higher the production rates achieved.
Good yield even for a large scale

Reaction kinetics
The second advantage of small channels over batch vessels is that the mixing and transport distances are reduced. With small-scale microreactors (channel sizes <500 µ), diffusion can be fast enough to mix reagents and the reactor channels can have simple, straight geometries (many reactor ‘chip’ or capillary based devices exist). However, if the size of a straight channel (or pipe) is increased beyond 1 mm, diffusion is too slow to mix the reagents effectively and the flow is laminar, meaning there is little cross channel motion. This results in poor mixing and different elements of the liquid travel along the channel at different rates – in other words, different parts of the reaction mixture spend different amounts of time in the reactor (poor plug flow). The serpentine shape of the reaction channel in the reactor plate is common throughout the Alfa Laval Plate Reactor range. It creates vortices in the liquid, which mix across the larger channels to ensure good and consistent plug flow behaviour; this characteristic facilitates more reliable and faster process scale-ups. In addition, the reaction channel is in close contact with the utility medium, which promotes good heat transfer. A range of accessories are available for multiple feeding sampling and instrumentation points, and the flexibility of the reactor design facilitates the application of process analytical technology (PAT).
Share: