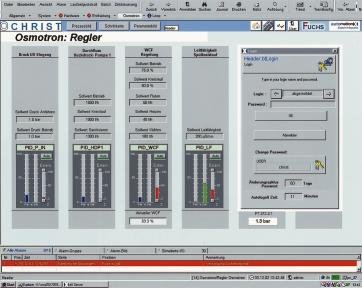
The FDA Regulation 21 CFR Part 11, which supports the introduction of paper-free recording systems, permits better process and plausibility checks, data integrity and protection against misuse, together with simple recognition of trends. The pure water specialist Christ therefore offers a customer-specific software and visualisation concept which complies with Part 11 instead of a convention- al stored-program controller (SPC). This is required, above all, wherever the pure water is used directly in the production process and/or for the US market. Such solutions demand an intensive exchange of infor-mation between the customer and the supplier. In the first phase, the Christ engineers define, in cooperation with the customer, the user interfaces of the automation classes to be developed. These are the basis for the individual software modules, which represent all physical and control components. The integrated control variant permits complete control of program modifications and the unrestricted documentation of all operator actions. Its control and representation function thus offers FDA-compliant recording of all product-quality parameters and all operator actions in an audit trail.
cpp 433
Share: